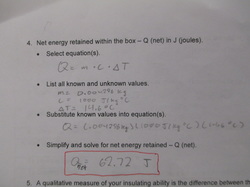Thermodynamics
Thermodynamics: The study of the effects of work, heat, flow, and energy on a system.
1st law of Thermodynamics: Energy cannot be created or destroyed. This one is pretty simple. It can't be destroyed but it changes from one form to another.
2nd law of Thermodynamics: Thermal energy flows from hot to cold. Basically when heat is radiated it goes from where it is hot and finds a way to cool off. An example of this is when you touch something cold and the heat from your hand tries to heat it up.
Zeroth's law of Thermodynamics: If two systems are separately found to be in thermal equilibrium with a third system, the first two are in equilibrium.
Entropy: Measure of how evenly heat is distributed within a system.
Three types of heat transfer:
Convection: Movement of thermal energy in a fluid. Basically it's how heat moves through air or a liquid.
Conduction: Heat from molecule to molecule. This is how heat is transferred by touching things.
Radiation: Where heat is transferred through electromagnetic waves. This is heat from light
U value: The meausre of a materials ability to conduct heat
R value: The measure of a materials ability to resist heat
-These two are the opposites of each other because one measure how good of an insulator one is and the other measures how good it conducts heat.
2nd law of Thermodynamics: Thermal energy flows from hot to cold. Basically when heat is radiated it goes from where it is hot and finds a way to cool off. An example of this is when you touch something cold and the heat from your hand tries to heat it up.
Zeroth's law of Thermodynamics: If two systems are separately found to be in thermal equilibrium with a third system, the first two are in equilibrium.
Entropy: Measure of how evenly heat is distributed within a system.
Three types of heat transfer:
Convection: Movement of thermal energy in a fluid. Basically it's how heat moves through air or a liquid.
Conduction: Heat from molecule to molecule. This is how heat is transferred by touching things.
Radiation: Where heat is transferred through electromagnetic waves. This is heat from light
U value: The meausre of a materials ability to conduct heat
R value: The measure of a materials ability to resist heat
-These two are the opposites of each other because one measure how good of an insulator one is and the other measures how good it conducts heat.
Equations and variable meanings
Q = Energy Transfer (joules)
m = mass of material (kilograms)
c = specific heat capacity (J/Kg C)
P = rate of energy transfer (Watts)
delta t = change in temperature (seconds)
k = thermal conductivity
A = area of thermal conductivity (meters)
L = thickness of material (meters)
delta T = difference in temperature
*delta is denoted as a triange
Q = m * c * delta T P = (k * A * delta T)/L k = (P * L)/ (A * delta T) P = Q/delta t
m = mass of material (kilograms)
c = specific heat capacity (J/Kg C)
P = rate of energy transfer (Watts)
delta t = change in temperature (seconds)
k = thermal conductivity
A = area of thermal conductivity (meters)
L = thickness of material (meters)
delta T = difference in temperature
*delta is denoted as a triange
Q = m * c * delta T P = (k * A * delta T)/L k = (P * L)/ (A * delta T) P = Q/delta t
You would use these equations to find how much energy or heat you lose in a system(Q) or what the rate of that energy transfer is (P) Engineers use these equations to find how well an insulator works by figuring out how much energy is lost in heat transfer (Q) You can also find the thermal conductivity of a certain material which tells you how well it absorbs heat (k) The specific heat capacity (c) can be found on the internet or you can calculate it if you run a heating lab on it like the one below to find the changes in temperature it undergoes, the mass of it and the energy transfer (Q)
Thermodynamics lab
In this lab we measured how heat escaped a small box with a lightbulb in it by putting an insulation that we made over it. We used a 1 inch thick slab of bubble wrap sandwiched in-between two pieces of cardboard. We used a Vernier temperature probe to calculate how ehat escaped over a 20 minute period after 20 minutes of heating.
Dimensions of the box: 0.00358 m^3
Light bulb wattage: 25 W
Initial internal temperature: 21 degrees celsius
Maximum internal temperature: 50.6 degreed celsius
Final internal temperature: 25.6 degrees celsius
Initial room temperature: 21.3 degrees celsius
Maximum room temperature: 23.2 degrees celsius
Final room temperature: 22.3 degrees celsius
Heating time: 20 minutes
Cooling time: 20 minutes
Density of air: 1.20 kg/m^3
Specific heat capacity of air: 1000 J/Kg C
Dimensions of the box: 0.00358 m^3
Light bulb wattage: 25 W
Initial internal temperature: 21 degrees celsius
Maximum internal temperature: 50.6 degreed celsius
Final internal temperature: 25.6 degrees celsius
Initial room temperature: 21.3 degrees celsius
Maximum room temperature: 23.2 degrees celsius
Final room temperature: 22.3 degrees celsius
Heating time: 20 minutes
Cooling time: 20 minutes
Density of air: 1.20 kg/m^3
Specific heat capacity of air: 1000 J/Kg C
Our qualitive measure of our insulators ability to retain heat is the difference between the maximum inside temperature and the maximum outside temperature. Our maximum internal temperature is 50.6 degrees celsius. Our maximum temperature on the outside was 23.2 degrees celsius, So the difference of those is 27.4 degrees celsius.
Conclusion:
Overall I thought doing the calculations for this lab was pretty fun and getting the end result was fun but the waiting 40 minutes to let the box heat up and cool down was the most boring part. Our box lost a little less than half of it's heat that it gained so I thought that was pretty good. This unit has given me a much better understanding of thermodynamics and how heat is transferred through a system.
Overall I thought doing the calculations for this lab was pretty fun and getting the end result was fun but the waiting 40 minutes to let the box heat up and cool down was the most boring part. Our box lost a little less than half of it's heat that it gained so I thought that was pretty good. This unit has given me a much better understanding of thermodynamics and how heat is transferred through a system.